biological abundance of elements
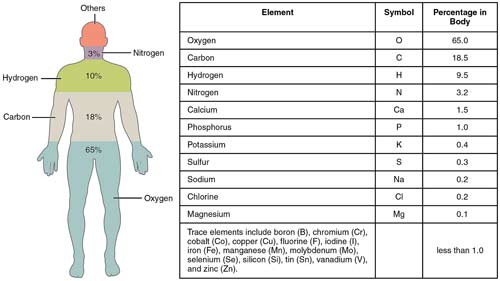
Element percentages in the human body.
In all terrestrial organisms, carbon, hydrogen, oxygen, and nitrogen are the primary constituents, making up 96.3% of the body mass of human beings and a similarly large proportion of all other organisms on Earth. Several factors contribute to their predominance: (a) They can combine with one another through covalent bonds to form molecules. (b) These bonds can be broken at temperatures compatible with life, so that the atoms can be rearranged into an enormous variety of other molecules. (c) Carbon is exceptional in its ability to form the basis of macromolecules. (d) Water, which is composed of oxygen and hydrogen atoms, plays a central role in the biochemistry of terrestrial life.
About 90% of the atoms (75% by weight) of the atoms in terrestrial organisms are oxygen and hydrogen. (e) Among the simpler molecules formed by the four main biological elements are water vapor and gases that are soluble in water. As water vapor in Earth's primordial atmosphere condensed and fell as rain, it brought the other dissolved substances into the early oceans. Subsequently, according to the consensus view, the interaction of small molecules led to more complex substances and eventually to life itself (see life, origin).
Interestingly, the four principal biological elements are also the four most common chemically active elements in the universe (see elements, cosmic abundance), helium and neon being inert. In contrast, only oxygen is highly abundant in both terrestrial organisms and the Earth's crust.
| Distribution of elements in the human body (by weight) | |||
|---|---|---|---|
| element | atomic no. | percentage | role |
| oxygen | 8 | 65.0 | cellular respiration, component of water |
| carbon | 6 | 18.5 | basis of organic molecules |
| hydrogen | 1 | 9.5 | component of water and most organic molecules, electron carrier |
| nitrogen | 7 | 3.3 | component of all proteins and nucleic acids |
| calcium | 20 | 1.5 | component of bones and teeth, triggers muscle contraction |
| phosphorus | 15 | 1.0 | component of nucleic acids, important in energy transfer |
| potassium | 19 | 0.4 | main positive ion inside cells, important in nerve function |
| sulfur | 16 | 0.3 | component of most proteins |
| sodium | 11 | 0.2 | main positive ion outside cells, important in nerve function |
| chlorine | 17 | 0.2 | main negative ion outside cells |
| magnesium | 12 | 0.1 | essential component of many energy-transferring enzymes |
| iron | 26 | trace | essential component of hemoglobin in the blood |
| copper | 29 | trace | component of many enzymes |
| molybdenum | 42 | trace | component of many enzymes |
| zinc | 30 | trace | component of some enzymes |
| iodine | 53 | trace | component of thyroid hormone |
Reference
1. Frieden, Earl. "The Chemical Elements of Life." Scientific American, July 1972, pp. 52–60.


